 The bond angle for this ion is 180 degrees as it has a linear geometry, and all the atoms are arranged in the same plane. From VSEPR theory, we know that this molecule would then be linear. Formal charge calculation enables you to. Looking at the arrangement of atoms and their valence electrons, it can be seen that the arrangement is planar, and as there are no lone pairs, there is no bent in its shape or geometry. This necessitates that a chemist knows the low energy electronic configuration for every one of the bonding atoms. Hence there is no lone pair around the C atom. O, N, and C are in groups 6, 5, and 4 and their. NCO- Molecular geometryĪs we already know, O is the most electronegative hence it should have the -1 formal charge, that there is a triple bond between N and C and a single bond exists between C and O. Draw a reasonable Lewis dot structure for ONC, including the formal charges (N.B. As a result, the Hybridization for the Carbon atom in NCO- ion is sp. And as there are two electron regions for Carbon, it forms two hybrid orbitals to form bonds with these two atoms.Ĭarbon forms one s-hybrid orbital and another p-hybrid orbital to form a triple bond and a single bond with the neighbouring atoms. Cabron forms a triple bond with the Nitrogen atom, which corresponds to one electron region, and it also forms a single bond with Oxygen, this is the second electron region. About Press Copyright Contact us Creators Advertise Developers Terms Privacy Policy & Safety How YouTube works Test new features Press Copyright Contact us Creators. Each electron counts as one and so a pair counts as two. 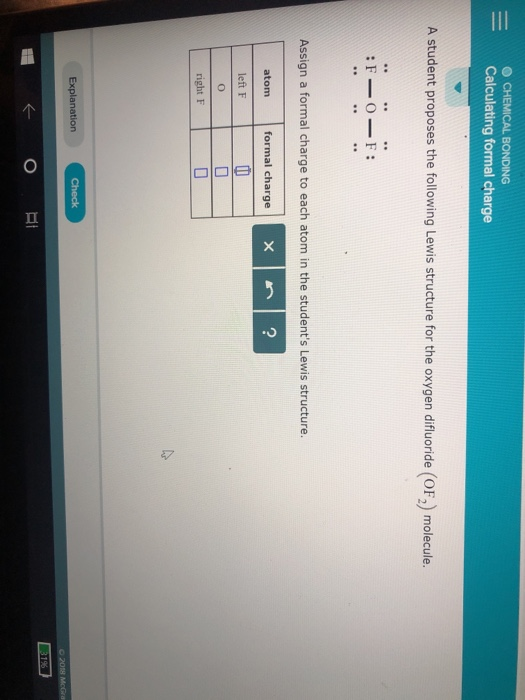
Lone Pairs lone electrons sitting on the atom. An online formal charge calculator is exclusively designed to calculate formal charge of an atom. Here as Carbon is forming bonds with Nitrogen and Oxygen atoms, we will look at its Hybridization. Formal Charge valence electrons on neutral atom ( lone electron pairs) + ( bonding electrons) Valence electrons corresponds to the group number of the periodic table (for representative elements). One of the easiest and simple ways to find out the Hybridization of any molecule is to look at the electron domain regions around the atom. However for Cyanate ion, the most accepted Lewis Structure if of that in which Carbon forms a triple bond with Nitrogen atom and a single bond with Oxygen atom. Compound interest is the formal name for the snowball effect in finance, where an initial amount grows upon itself and gains more and more momentum over time. Alternative Method Count up the number of electrons in the atoms 'circle.' Since the circle cuts the covalent bond 'in half,' each. FC (Number of valence electrons)-(Number of bonding. 
The above Lewis structures indicate that the cyanate ion is capable of bonding at either the Nitrogen or Oxygen to a metal atom. The FC is the charge an atom would have if all atoms in the molecule had the same electronegativity. This suggests that option A is the correct answer. Therefore, the Lewis structures for NCO- are as follows: The formal charges on the nitrogen atoms in the case of azide ion from left to right are -1, +1, -1. Delocalization electron pairs draw all the other equivalent resonance structures. 
Unshared electron pairs are added so that there is an octet of electrons around each atom. 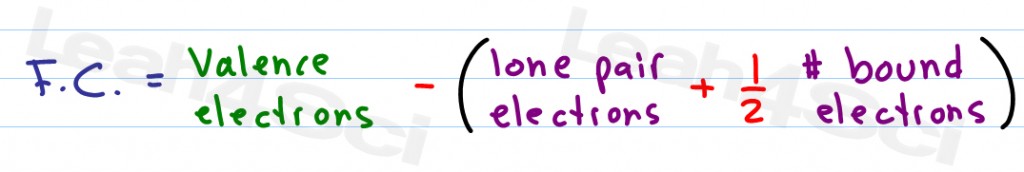
Alternatively, one triple bond is added either between C and O or between C, and N. Calculating Formal Charge: The following equation determines the formal charge for each atom in a molecule or polyatomic ion. Show a single bond between the central atom is Carbon, with its neighbouring atoms ( Oxygen and Nitrogen).Ī double bond is added between C and O in the structure, and a second double bond is added between C and N. The central atom for Cyanate will be the C atom since it is the least electronegative. Here as we already know that there are 16 valence electrons, we can determine the central atom for this ion.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
